
Центр коллективного пользования «СКИФ» (Сибирский кольцевой источник фотонов)
На Общем собрании СО РАН директор Института катализа им. Г.К. Борескова СО РАН академик Валерий Иванович Бухтияров рассказал о том, какими будут первые шесть станций нового центра коллективного пользования «СКИФ», создаваемого в рамках проекта «Академгородок 2.0», и какие исследования на них планируется проводить.

Новосибирский «СКИФ» станет частью сетевой российской инфраструктуры синхротронных исследований нового поколения, куда помимо него войдет головной объект на базе НИЦ «Курчатовский институт» в г. Протвино Московской области, а также специализированный объект во Владивостоке на острове Русский. «СКИФ» относится к четвертому поколению приборов, его энергия составит 3 ГэВ, а эмиттанс – 186-60, что позволит проводить исследования на живых объектах. «Эти исследования улучшат возможности Сибирского отделения в фундаментальных и прикладных работах», – говорит Валерий Бухтияров.
Всего запланировано 32 экспериментальные станции, в рамках первой очереди будут построены шесть: сканирующего микроанализа – «Микрофокус»; структурной диагностики; исследования быстропротекающих процессов; XAFS-спектроскопии и магнитного дихроизма; диагностики в высокоэнергетическом рентгеновском диапазоне; электронной структуры.
Станция «Микрофокус» будет сочетать сверхмалый эмиттанс и достижения рентгеновской оптики. На этой станции планируется решать несколько блоков задач. Во-первых, изучать структуру биополимеров, в том числе исследовать систему репарации человека, передачу наследственной информации, причины и механизмы заболеваний для создания лекарственных препаратов. Во-вторых, работать в сфере создания новых материалов, то есть изучать структуру и другие показатели в многокомпонентных веществах, смотреть, как ведут себя новые материалы в разных условиях давления, температур и излучений разного спектра. Наконец, станция «Микрофокус» позволит проводить исследования геоматериалов, то есть выявлять компоненты рудных ископаемых, проводить эксперименты при высоких давлениях и температурах, чтобы понять геологические процессы и подобрать оптимальные условия для синтеза искусственных минералов.
Станция структурной диагностики позволит исследовать объекты методами рентгеновской дифракции. Она найдет применение в сфере катализаторов для нефтепереработки и экологических приложений, а также для материалов различных назначений, например, тонких пленок, систем с частично разупорядоченной иерархической структурой, наноматериалов, монокристаллов, фармацевтических материалов и других.
Станция исследования быстропротекающих процессов направлена на изучение кристаллической структуры вещества в экстремальных условиях высоких температур (10000°C) и давлений (10 млн атм), образующихся при воздействии на него ударных, детонационных волн, мощного лазерного излучения, плазмы и др. Она будет включать в себя три секции: динамических процессов, плазмы и экстремально высоких температур.
Станция XAFS-спектроскопии (Х-ray absorption fine structure – тонкая структура спектров поглощения рентгеновских лучей – Прим. ред.) и магнитного дихроизма будет применяться для исследования электронной и локальной структуры веществ. Например, с ее помощью можно посмотреть центры связывания металлов в белках или процессы осаждения и роста полупроводниковых пленок.
Станция диагностики в высокоэнергетическом рентгеновском диапазоне позволит получить контрастные изображения высокого разрешения и в реальном времени в жестком рентгеновском диапазоне. Например, с ее помощью можно изучать палеонтологические находки, новые композитные материалы и геологические образцы, а также получать их трехмерное изображения.
Станция электронной структуры позволит исследовать электронную структуру в разных ситуациях: на поверхности, в объеме и на границах раздела фаз в разных материалах, пленках и многослойных покрытиях твердых тел.
Валерий Бухтияров отметил, что
сейчас идет процесс обсуждения станций второй очереди: планируется
исследовать структуру и форму вирусов методом рентгеновской
дифракции, изучать электронное строение, структуру и состав веществ
различной природы, а также превращение веществ при электронно-лучевой
и
лазерной сварке, и при воздействии потоков частиц на
поверхность. Кроме того, намечаются работы по изучению того, что
происходит на первой стенке термоядерного реактора при облучении
потоками плазмы (сегодня
создание первой стенки термоядерного реактора ITER — одна из
наиболее актуальных проблем – Прим. ред.)
и исследования электронной и спиновой структуры кристаллов.
Газета “Наука в Сибири”
и сайт Института катализа
24-25 сентября 2018 г., Томск, Россия
http://www.catalysis.ru/resources/science/conferences/newcat_2018/index.html
С 24 по 25 сентября 2018 года в Малом зале Научной библиотеки Томского государственного университета прошла Вторая школа молодых учёных «Новые каталитические процессы глубокой переработки углеводородного сырья и биомассы». Организаторами школы выступили Институт катализа им. Г.К. Борескова СО РАН, Российский научный фонд, Национальный исследовательский Томский государственный университет и АО «Газпромнефть - Омский нефтеперерабатывающий завод». Школа проводится ежегодно в рамках выполнения проекта Российского научного фонда.

В работе школы приняли участие около 60 ведущих специалистов и молодых ученых, среди которых было 11 пленарных лекторов и 50 слушателей из Новосибирска, Томска, Красноярска, Омска, Твери, Иваново, Казани, Москвы, Кемерово и Санкт-Петербурга.

Открывал пленарную сессию председатель школы академик РАН Валентин Николаевич Пармон (Институт катализа СО РАН), лауреат премии «Глобальная энергия» за 2016 год. Валентин Николаевич прочитал лекцию «Каталитические процессы в условиях сильных физических воздействий», раскрыв суть протекающих процессов на наиболее ярких практических примерах, таких как термокаталитические циклы «ЕВА-АДАМ» и «ИКАР» для конверсии ядерной и солнечной энергии в химическую. Суть процессов заключается в преобразовании первичной формы энергии в химическую с помощью обратимых химических реакций термокаталитических циклов либо квантового (фотокаталитического) преобразования энергии.
Термокаталитический цикл «ЕВА-АДАМ» разрабатывался для сглаживания суточной нагрузки на ядерный реактор путем конверсии ядерной энергии в химическую. В цикле «ЕВА-АДАМ» запасание энергии происходит в смеси СО и Н2, получаемой в паровой конверсии метана в синтез-газ, а выделение энергии – путем метанирования синтез-газа до метана и воды. Основным недостатком цикла «ЕВА-АДАМ» является малая удельная энергонапряженность (УЭН) установок с теплообменниками, составляющая всего 1/40-1/20 от УЭН ядерного реактора. Особенностью цикла «ИКАР» (разработка ИК СО РАН) является подвод энергии к катализатору в виде ионизирующего излучения без промежуточных теплообменников, что обеспечивает повышение УЭН в 40 раз по сравнению с циклом «ЕВА-АДАМ». В.Н. Пармон отметил, что возможность высокоэффективного преобразования солнечной энергии в химическую была экспериментально подтверждена при испытаниях опытных установок разной мощности. К примеру, к.п.д. опытной установки с полезной мощностью около 2 кВт достигал 43% для преобразования солнечной энергии и 20% – для полного цикла аккумулирование-выделение энергии. Достоинством фотокаталитических методов преобразования является отсутствие необходимости предварительной концентрации солнечного света. В лекции были также рассмотрены вопросы инициирования процессов каталитического превращения тяжелых углеводородных нефтяных остатков при высокоинтенсивном воздействии на них ионизирующим и электромагнитным излучением.
 Следующим выступил заместитель директора по научной работе Института углехимии и химического материаловедения Федерального исследовательского центра угля и углехимии СО РАН к.х.н. Алексей Петрович Козлов
– специалист в области углехимии и химии углеродных материалов. Алексей Петрович прочитал пленарную лекцию «Научные основы производства игольчатого кокса и углеродных сорбентов из нефтяного сырья и из ископаемых углей».
Он рассказал, что игольчатый кокс это высокотекстурированный углеродный материал, используемый для получения крупногабаритных графитированных электродов, применяемых при выплавке стали в электродуговых печах. Основным условием получения
высококачественного игольчатого кокса является квалифицированный подбор и подготовка исходного сырья коксования. В качестве сырья традиционно используются малосернистые остатки термодеструктивного происхождения: дистиллятные крекинг-остатки, смолы пиролиза, тяжелые
газойли каталитического крекинга с современных установок FCC.
Следующим выступил заместитель директора по научной работе Института углехимии и химического материаловедения Федерального исследовательского центра угля и углехимии СО РАН к.х.н. Алексей Петрович Козлов
– специалист в области углехимии и химии углеродных материалов. Алексей Петрович прочитал пленарную лекцию «Научные основы производства игольчатого кокса и углеродных сорбентов из нефтяного сырья и из ископаемых углей».
Он рассказал, что игольчатый кокс это высокотекстурированный углеродный материал, используемый для получения крупногабаритных графитированных электродов, применяемых при выплавке стали в электродуговых печах. Основным условием получения
высококачественного игольчатого кокса является квалифицированный подбор и подготовка исходного сырья коксования. В качестве сырья традиционно используются малосернистые остатки термодеструктивного происхождения: дистиллятные крекинг-остатки, смолы пиролиза, тяжелые
газойли каталитического крекинга с современных установок FCC.
В лекции было отмечено, что Россия располагает огромными запасами каменных углей битуминозного типа (Ж, Д, Г, ГЖ, ОС, КС) в Кузбассе. Также в районе Кузбасса имеются мощные производства каменноугольных пеков. Поэтому именно на этой сырьевой базе и была начата разработка отечественной углепековой технологии получения активных углей с использованием метода парогазовой активации, что безусловно даст мощный толчок развитию производительных сил и обеспечению эффективной защиты окружающей среды.
Лекцию «Катализаторы для гидрогенизационных процессов переработки нефтяных фракций: существующий уровень и перспективы развития» прочитал заведующий Лабораторией катализаторов нефтепереработки Института катализа СО РАН к.х.н. Олег Владимирович Климов. Гидрогенизационные каталитические процессы развиваются опережающими темпами по сравнению с другими группами процессов нефтепереработки. Причинами этого являются как повышенные выходы целевых светлых нефтепродуктов, так и их качество, удовлетворяющее современным и перспективным стандартам, недостижимые без использования гидрогенизационных процессов.
 Постоянное возрастание экологических и химмотологических требований к качеству получаемых нефтепродуктов; необходимость увеличения выходов целевых
продуктов; постоянное увеличение глубины переработки нефти, обуславливающее необходимость вовлечения в переработку все больших количеств труднопревращаемых вторичных фракций; необходимость снижения энергоёмкости гидрогенизационных процессов – все
вышеперечисленные факторы приводят к тому, что происходит постоянное совершенствование катализаторов гидрогенизационных процессов.
Постоянное возрастание экологических и химмотологических требований к качеству получаемых нефтепродуктов; необходимость увеличения выходов целевых
продуктов; постоянное увеличение глубины переработки нефти, обуславливающее необходимость вовлечения в переработку все больших количеств труднопревращаемых вторичных фракций; необходимость снижения энергоёмкости гидрогенизационных процессов – все
вышеперечисленные факторы приводят к тому, что происходит постоянное совершенствование катализаторов гидрогенизационных процессов.
В лекции были рассмотрены современные отечественные катализаторы гидроочистки дизельного топлива, вакуумного газойля и бензина каталитического крекинга, а также катализаторы первой и второй стадий гидрокрекинга вакуумного газойля, проведено сопоставление эксплуатационных характеристик этих катализаторов с промышленными образцами ведущих мировых производителей и определены основные направления дальнейшего совершенствования катализаторов.
 Продолжила пленарную сессию лекция «Роль каталитических процессов в ключевых стадиях промышленного синтеза капролактама», представленная ведущим научным сотрудником лаборатории каталитических исследований Томского
государственного университета д.х.н. Эльдаром Мусаевичем Дахнави. Несмотря на некоторые достижения в области усовершенствования, производство капролактама все еще остается очень энергоемким с высокими расходными
нормами сырья и c образованием большего количества побочных и сопутствующих продуктов. Все российские производственные мощности по капролактаму базируются на технологии промежуточного образования циклогексанона (Анон) из циклогексана (ЦГ), с его дальнейшим
оксимированием и перегруппировкой получаемого оксима в капролактам. Общий выход капролактама на поданный бензол, в среднем, составляет около 70 %, что свидетельствует о невысоком уровне данного промышленного процесса. Наиболее «слабым звеном» в
рассматриваемой технологической цепочке является стадия окисления циклогексана, и для увеличения эффективности всего производства необходимо улучшить показатели синтеза и выделения Анона из реакционной массы.
Продолжила пленарную сессию лекция «Роль каталитических процессов в ключевых стадиях промышленного синтеза капролактама», представленная ведущим научным сотрудником лаборатории каталитических исследований Томского
государственного университета д.х.н. Эльдаром Мусаевичем Дахнави. Несмотря на некоторые достижения в области усовершенствования, производство капролактама все еще остается очень энергоемким с высокими расходными
нормами сырья и c образованием большего количества побочных и сопутствующих продуктов. Все российские производственные мощности по капролактаму базируются на технологии промежуточного образования циклогексанона (Анон) из циклогексана (ЦГ), с его дальнейшим
оксимированием и перегруппировкой получаемого оксима в капролактам. Общий выход капролактама на поданный бензол, в среднем, составляет около 70 %, что свидетельствует о невысоком уровне данного промышленного процесса. Наиболее «слабым звеном» в
рассматриваемой технологической цепочке является стадия окисления циклогексана, и для увеличения эффективности всего производства необходимо улучшить показатели синтеза и выделения Анона из реакционной массы.
В процессе окисления циклогексана в качестве катализатора, в основном, используется нафтенат кобальта. Основная проблема, возникающая при использовании данного катализатора, заключается в том, что в условиях промышленного процесса он выпадает в осадок в виде адипината кобальта с дальнейшим осмолением стенок реакторов и трубопроводов, а целевые продукты (Анол, Анон и ГПЦГ) образуются в «неудобных» соотношениях для получения Анона с максимальным выходом. По этой причине ведущие мировые производители капролактама отказались от нафтената кобальта или вовсе перешли на некаталитическое окисление циклогексана. В настоящее время разрабатываются различные катализаторы селективного окисления циклогексана, наиболее перспективными являются бинарные каталитические системы. В их присутствии конверсия циклогексана за проход достигается более 10 % с обеспечением селективности по целевым продуктам на уровне 90 % ÷ 95 %, что вселяет надежду на коренные улучшения производства капролактама.
В лекции было отмечено, что замена одного катализатора на другой не решает существующие проблемы производства, для их успешного решения такая замена должна сопровождаться комплексным использованием новых инжиниринговых подходов. Большое внимание было уделено результатам лабораторных исследований и вопросам селективного окисления циклогексана в присутствии различных каталитических систем, моделированию и масштабированию ключевых стадий, а также поиску путей достижения высоких показателей производства капролактама.
 Далее выступил ведущий научный сотрудник Лаборатории каталитических методов преобразования солнечной энергии Института катализа СО
РАН, д.х.н. Владимир Викторович Чесноков – специалист в области механизмов образования углерода из углеводородов на катализаторах различной природы, синтеза углеродных наноматериалов, новых каталитических
процессов глубокой переработки углеводородного сырья. Лекция «Каталитические методы обезвреживания хлорпроизводных углеводородов» была посвящена проблеме утилизации или уничтожения отходов, содержащих
хлорпроизводные углеводороды. Актуальность данной темы обусловлена в первую очередь необходимостью защиты окружающей среды от вредных промышленных отходов. Исследование новых каталитических систем в
данной области могло бы позволить разработать новый способ разложения хлоруглеводородов не только с целью утилизации, но и для получения из них ценных химических продуктов. В лекции были
представлены результаты работы по разработке каталитических методов обезвреживания хлорпроизводных углеводородов.
Далее выступил ведущий научный сотрудник Лаборатории каталитических методов преобразования солнечной энергии Института катализа СО
РАН, д.х.н. Владимир Викторович Чесноков – специалист в области механизмов образования углерода из углеводородов на катализаторах различной природы, синтеза углеродных наноматериалов, новых каталитических
процессов глубокой переработки углеводородного сырья. Лекция «Каталитические методы обезвреживания хлорпроизводных углеводородов» была посвящена проблеме утилизации или уничтожения отходов, содержащих
хлорпроизводные углеводороды. Актуальность данной темы обусловлена в первую очередь необходимостью защиты окружающей среды от вредных промышленных отходов. Исследование новых каталитических систем в
данной области могло бы позволить разработать новый способ разложения хлоруглеводородов не только с целью утилизации, но и для получения из них ценных химических продуктов. В лекции были
представлены результаты работы по разработке каталитических методов обезвреживания хлорпроизводных углеводородов.
Опираясь на знания о механизме «карбидного цикла» образования углеродных нановолокон (УНВ), был разработан метод обезвреживания хлорпроизводных углеводородов. Было установлено, что переработку хлоруглеводородов и их смесей на катализаторах, содержащих металлы подгруппы железа, возможно осуществлять в двух различных технологических режимах: 1) в режиме разложения по механизму «карбидного цикла» до УНВ и HCl (в условиях стехиометрического количества Н2) и 2) в режиме гидродехлорирования (в условиях избытка Н2) с получением HCl и смеси углеводородов, не содержащих хлор. Показана применимость метода для утилизации как алифатических, так и ароматических хлорпроизводных углеводородов.
 Первый рабочий день школы молодых ученых завершала лекция «Сжигание в кипящем слое катализатора – универсальная технология
переработки органического сырья», прочитанная к.х.н. Юрием Владимировичем Дубининым – научным сотрудником Лаборатории каталитических процессов переработки возобновляемого сырья Института катализа СО РАН. По наблюдениям
докладчика, общим недостатком всех традиционных способов сжигания топлива является высокая температура процесса, что приводит к загрязнению атмосферы токсичными продуктами сгорания. Кроме того, в
настоящее время большой интерес вызывает включение в топливные ресурсы не использовавшихся ранее низкокачественных топлив, в том числе техногенных отходов и возобновляемой биомассы. Использование таких материалов сильно затруднено или невозможно вследствие их
свойств (низкая калорийность, высокая влажность) и высокой экологической опасности процесса их переработки. Таким образом, необходима современная технология утилизации различного рода топлив и отходов, сочетающая в себе высокую эффективность и соответствие всем
существующим экологическим стандартам.
Первый рабочий день школы молодых ученых завершала лекция «Сжигание в кипящем слое катализатора – универсальная технология
переработки органического сырья», прочитанная к.х.н. Юрием Владимировичем Дубининым – научным сотрудником Лаборатории каталитических процессов переработки возобновляемого сырья Института катализа СО РАН. По наблюдениям
докладчика, общим недостатком всех традиционных способов сжигания топлива является высокая температура процесса, что приводит к загрязнению атмосферы токсичными продуктами сгорания. Кроме того, в
настоящее время большой интерес вызывает включение в топливные ресурсы не использовавшихся ранее низкокачественных топлив, в том числе техногенных отходов и возобновляемой биомассы. Использование таких материалов сильно затруднено или невозможно вследствие их
свойств (низкая калорийность, высокая влажность) и высокой экологической опасности процесса их переработки. Таким образом, необходима современная технология утилизации различного рода топлив и отходов, сочетающая в себе высокую эффективность и соответствие всем
существующим экологическим стандартам.
Для решения этой проблемы в Институте катализа СО РАН разработан нетрадиционный способ сжигания жидких, газообразных и твердых топлив в присутствии катализаторов, который позволяет ликвидировать многие недостатки высокотемпературного сжигания топлив. Каталитическое горение принципиально отличается от горения в традиционном понимании, так как органические вещества окисляются на поверхности твердых катализаторов без образования пламени.
Метод низкотемпературного каталитического окисления различных топлив и отходов в псевдоожиженном слое позволяет исключить большинство недостатков традиционных способов сжигания. В том числе: проводить процесс в автотермическом режиме, т.е. без затрат дополнительного топлива; снизить габариты и металлоемкость аппаратов более чем в 15 раз; ликвидировать или резко уменьшить образование газовых выбросов, загрязненных токсичными органическими веществами, в том числе диоксинами, оксидами углерода, азота и серы.
Технология сжигания в кипящем слое катализатора была успешно применена на пяти различных котельных мощностью от 1 до 6 Гкал/час и зарекомендовала себя как эффективный и экологически безопасный способ получения тепла.
 Второй рабочий день мероприятия был открыт лекцией «Процессы и катализаторы конверсии природных углеводородных газов в практически
важные продукты». Докладчиком выступил директор Института химии нефти СО РАН, специалист в области нефтехимии, катализа, газо- и нефтепереработки д.х.н. Александр Владимирович Восмериков.
Работы А.В. Восмерикова посвящены решению одной из важнейших народно-хозяйственных задач – разработке и созданию высокоэффективных катализаторов для процессов переработки углеводородного сырья. В лекции обсуждались вопросы
переработки природного газа, считающегося перспективным источником энергии на фоне острой необходимости замены нефти на альтернативные источники углеводородов.
Второй рабочий день мероприятия был открыт лекцией «Процессы и катализаторы конверсии природных углеводородных газов в практически
важные продукты». Докладчиком выступил директор Института химии нефти СО РАН, специалист в области нефтехимии, катализа, газо- и нефтепереработки д.х.н. Александр Владимирович Восмериков.
Работы А.В. Восмерикова посвящены решению одной из важнейших народно-хозяйственных задач – разработке и созданию высокоэффективных катализаторов для процессов переработки углеводородного сырья. В лекции обсуждались вопросы
переработки природного газа, считающегося перспективным источником энергии на фоне острой необходимости замены нефти на альтернативные источники углеводородов.
Сегодня природный газ в основном используется как топливо. Однако потенциальная его ценность как химического сырья существенно выше, чем как энергоносителя. Он может служить базой для производства большинства органических соединений, получаемых при переработке нефти. В то же время современные методы его переработки оказываются недостаточно эффективными для реализации этого потенциала. Другим перспективным источником углеводородов является попутный нефтяной газ (ПНГ). Помимо метана он содержит широкую фракцию легких углеводородов – смесь этана, пропана, бутанов, пентанов и т.д., поэтому рассматривается как важный ресурс для газохимии.
Перспективным направлением рационального использования природных углеводородных газов представляется одностадийный каталитический синтез из них ароматических соединений. Процесс получения жидких продуктов из углеводородных газов протекает на цеолитсодержащих катализаторах и включает ряд параллельных и последовательных реакций: крекинга, дегидрирования, олигомеризации, диспропорционирования, циклизации и другие.
Состав получаемых продуктов, а также закономерности его изменения в зависимости от условий проведения реакции указывают на то, что образование ароматических углеводородов проходит через несколько стадий. На первой стадии протекают реакции крекинга и дегидрирования, приводящие к образованию стабильных продуктов – метана, этана и водорода, а также низших олефинов, которые вступают в дальнейшие превращения с образованием ароматических углеводородов и поликонденсированных соединений (кокса).
Низшие олефиновые мономеры – этилен и пропилен – относятся к числу наиболее важных и востребованных рынком химических продуктов. Метод пиролиза, которым традиционно получают основное количество низших олефинов, обладает целым рядом существенных недостатков. Альтернативным способом получения низших олефинов является превращение газообразных углеводородов на катализаторах, приготовленных на основе цеолитов различного структурного типа. Проведение процессов в их присутствии позволяет достигать более высокого выхода целевого продукта, при этом температура проведения реакции, как правило, существенно ниже, чем для традиционного пиролиза. Модификация цеолитов позволяет существенно повысить выход целевого продукта и снизить образование нежелательных продуктов.
В лекции был сделан вывод о том, что наиболее перспективными направлениями использования низших алканов являются как комплексное получение одновременно смеси ароматических углеводородов, бензола и сухого газа, так и этилена с пропиленом. Промышленная реализация способов каталитической переработки природных углеводородных газов обеспечит не только высокий экономический эффект и способствует удовлетворению растущих потребностей рынка в указанных продуктах, но также приведет к улучшению экологической обстановки в регионах их добычи и производства.
 Далее выступила к.х.н. Светлана Анатольевна Яшник (старший научный сотрудник Лаборатории экологического катализа Института
катализа СО РАН) с лекцией «Подходы к регулированию каталитических свойств Cu-замещенных цеолитов в пероксидном окислении органических экотоксикантов и функционализации метана».
Медь-замещенные цеолиты интенсивно изучаются в силу широкого спектра их практического применения, включающего как процессы очистки газовых выбросов и водных стоков от экотоксикантов, так и синтеза
востребованных органических соединений. Каталитические характеристики Cu-содержащих цеолитов в реакциях глубокого и селективного окисления органических соединений контролируются окислительными свойствами
Cu-структур, зависящими от электронного состояния иона меди, наличия в Cu-структурах реакционноспособного кислорода или способности к его активации, взаимодействия Cu-структур с каркасом цеолита, условиями процесса.
Далее выступила к.х.н. Светлана Анатольевна Яшник (старший научный сотрудник Лаборатории экологического катализа Института
катализа СО РАН) с лекцией «Подходы к регулированию каталитических свойств Cu-замещенных цеолитов в пероксидном окислении органических экотоксикантов и функционализации метана».
Медь-замещенные цеолиты интенсивно изучаются в силу широкого спектра их практического применения, включающего как процессы очистки газовых выбросов и водных стоков от экотоксикантов, так и синтеза
востребованных органических соединений. Каталитические характеристики Cu-содержащих цеолитов в реакциях глубокого и селективного окисления органических соединений контролируются окислительными свойствами
Cu-структур, зависящими от электронного состояния иона меди, наличия в Cu-структурах реакционноспособного кислорода или способности к его активации, взаимодействия Cu-структур с каркасом цеолита, условиями процесса.
Представленные в лекции подходы к регулированию каталитических свойств Cu-замещенных цеолитов основаны, преимущественно, на химических аспектах регулирования структуры медьсодержащих центров в ходе синтеза Cu-цеолитов и экспериментальной информации о взаимосвязи структуры центров с их свойствами.
Многообразие структурных типов цеолита, различающихся пространственной организацией и размером каналов, а также химический состав каркаса цеолита и размер его кристаллитов (для одного структурного типа) определяют специфичность взаимодействия комплексов меди с обменными ионами цеолита, кислотные свойства, доступность формирующихся структур меди для реагентов, а, следовательно, дают еще одну возможность регулирования функциональных свойств Cu-цеолитов. Выбор оптимальных реакционных условий, в частности рН среды, тоже позволяет контролировать окислительные свойства Cu-замещенных цеолитов.
 Старший научный сотрудник Лаборатории каталитических методов преобразования солнечной энергии Института
катализа СО РАН к.х.н. Дмитрий Александрович Булушев продолжил научную программу школы с выступлением на тему «Получение водорода из муравьиной кислоты на гетерогенных катализаторах».
Получение водорода из муравьиной кислоты вызывает особый интерес в связи с тем, что эта кислота может быть использована для хранения водорода и получена из биомассы или гидрированием СО2
при низких температурах. Было рассмотрено газофазное разложение муравьиной кислоты. Для этой реакции необходимы катализаторы и условия, которые определяют высокие скорости и селективности процесса
по отношению к водороду и CO2. Перспективными носителями являются углеродные пористые материалы, поскольку их поверхность высока и они инертны по отношению к кислым и щелочным средам. Кроме того, имеется возможность их допирования
кислородом или азотом с целью изменения активности катализатора.
Старший научный сотрудник Лаборатории каталитических методов преобразования солнечной энергии Института
катализа СО РАН к.х.н. Дмитрий Александрович Булушев продолжил научную программу школы с выступлением на тему «Получение водорода из муравьиной кислоты на гетерогенных катализаторах».
Получение водорода из муравьиной кислоты вызывает особый интерес в связи с тем, что эта кислота может быть использована для хранения водорода и получена из биомассы или гидрированием СО2
при низких температурах. Было рассмотрено газофазное разложение муравьиной кислоты. Для этой реакции необходимы катализаторы и условия, которые определяют высокие скорости и селективности процесса
по отношению к водороду и CO2. Перспективными носителями являются углеродные пористые материалы, поскольку их поверхность высока и они инертны по отношению к кислым и щелочным средам. Кроме того, имеется возможность их допирования
кислородом или азотом с целью изменения активности катализатора.
В работе было рассмотрено влияние природы активного металла и носителей катализаторов на каталитические свойства. Предложены пути увеличения активности и селективности через допирование углеродного носителя азотом, а катализаторов – ионами щелочного металла. Рассмотрены механизмы допирования.
Муравьиная кислота также может быть использована в качестве донора водорода вместо молекулярного водорода для различных реакций. Хранение кислоты, ее транспортировка и использование не требуют таких сложных подходов, как для молекулярного водорода. В докладе была также показана возможность применения муравьиной кислоты для синтезатоплив.
 Лекция «Получение малотоннажных химических продуктов и биотоплив из древесины» была прочитана заведующим лабораторией комплексной переработки биомассы
Института химии и химической технологии СО РАН (Красноярск) д.х.н. Валерием Евгеньевичем Тарабанько, в основную сферу интересов которого входят исследование топлив из возобновляемого растительного сырья; химия древесины; исследование
процессов переработки углеводов и лигнинов в ценные химические продукты. Цель доклада заключалась в оценке современного состояния исследований и перспектив развития процессов переработки растительного сырья в биотоплива, а также такие продукты как
фурфурол, левулиновая кислота и другие продукты тонкого органического синтеза.
Лекция «Получение малотоннажных химических продуктов и биотоплив из древесины» была прочитана заведующим лабораторией комплексной переработки биомассы
Института химии и химической технологии СО РАН (Красноярск) д.х.н. Валерием Евгеньевичем Тарабанько, в основную сферу интересов которого входят исследование топлив из возобновляемого растительного сырья; химия древесины; исследование
процессов переработки углеводов и лигнинов в ценные химические продукты. Цель доклада заключалась в оценке современного состояния исследований и перспектив развития процессов переработки растительного сырья в биотоплива, а также такие продукты как
фурфурол, левулиновая кислота и другие продукты тонкого органического синтеза.
Очевидно, что применение даже самых простых технологий переработки растительного сырья в биотоплива сдерживается их высокой стоимостью по сравнению с ценами нефтепродуктов и других традиционных энергоносителей. Вторая почти столь же очевидная проблема – масштаб необходимых ресурсов сырья. С этих позиций перспективны процессы переработки древесины в более дорогие малотоннажные продукты. Среди них можно отметить такие традиционные продукты, как ванилин и сиреневый альдегид из лигнина, левулиновую кислоту из целлюлозы и фурфурол из пентозных гемицеллюлоз. За исключением ванилина, получение этих продуктов целесообразно только из древесины или иного растительного сырья. Другая возможность переработки древесины, сочетающая моторные топлива в качестве цели и высокую рентабельность процессов – получение дорогих топливных присадок, синтез которых из нефти может быть менее экономичен. Среди них можно отметить этилфурфуриловый эфир и фурфуриламин, разветвленные углеводороды для зимнего дизельного топлива и др.
Основная часть лекции была посвящена развитию новых эффективных методов прямой, двухступенчатой переработки древесины в перечисленные выше ванилин, сиреневый альдегид, левулиновую кислоту и фурфурол. Принципиальный результат проведенных исследований состоит в том, что найдены компромиссные условия получения и ванилина, и целлюлозы в каталитическом процессе окислительной делигнификации древесины. На примере одного и того же ферментного комплекса показано, что целлюлоза, продукт разработанного процесса окисления древесины в ванилин, гидролизуется в глюкозу намного легче по сравнению как с исходной древесиной, так и промышленными видами целлюлозы.
 Продолжила тему биопереработки старший научный сотрудник Лаборатории каталитических методов преобразования солнечной энергии Института катализа СО РАН к.б.н.
Ксения Николаевна Сорокина с лекцией «Перспективные источники возобновляемого сырья и их биотехнологическая переработка в востребованные продукты».
Продолжила тему биопереработки старший научный сотрудник Лаборатории каталитических методов преобразования солнечной энергии Института катализа СО РАН к.б.н.
Ксения Николаевна Сорокина с лекцией «Перспективные источники возобновляемого сырья и их биотехнологическая переработка в востребованные продукты».
В связи с увеличением в последние десятилетия мирового энергопотребления и ограниченностью запасов природного ископаемого сырья, возникает потребность в использовании новых возобновляемых источников энергии. В числе перспективных видов сырья для получения биотоплива и химических веществ в настоящее время используются энергетические сельскохозяйственные культуры, а также продукты и отходы переработки биомассы.
Для получения сахаров используется ряд подходов, позволяющих эффективно гидролизовать предварительно активированную растительную биомассу с использованием как химических, так и биологических катализаторов (ферментов целлюлаз). В работе были рассмотрены свойства различных биотехнологических методов ферментации деполимеризованной биомассы. Результатом ферементации является получение ряда веществ, применяющихся в качестве биотоплива (этанол, н-бутанол, изобутанол), а также химических веществ (2,3-бутандиол, молочная и янтарная кислоты). Показано, что подход, связанный с консолидированной переработкой биомассы (CBP) в востребованные вещества является перспективным для прямого получения химических веществ путем ферментации биомассы, но пока уступает другим способам переработки по свой эффективности.
Другим перспективным видом возобновляемого сырья являются микроводоросли. Этот источник сырья обладает большой скоростью воспроизводства и способностью накапливать значительное количество высокоэнергетических липидов за счет высокой фотосинтетической активности. Несмотря на высокий уровень накопления липидов микроводорослями, производство биодизельного топлива на их основе пока остается нерентабельным из-за высокой стоимости получения липидов. Применение как химических, так и биологических катализаторов обеспечивает высокий выход метиловых эфиров жирных кислот из масла микроводорослей. Другим перспективным продуктом, получаемым из биомассы микроводорослей, являются углеводы (в том числе крахмал). Обработка биомассы микроводорослей с использованием ферментов или прямая ферментация углеводов с использованием микроорганизмов позволяет получать этанол, также применяющийся в качестве биотоплива.
Развитие методов генетической инженерии микроорганизмов и применение методов системной биологии в будущем приведут к разработке более совершенных биотехнологических процессов, позволяющих получать биотопливо и мономеры для химического синтеза.

В 2018 году школа молодых ученых «Новые каталитические процессы глубокой переработки углеводородного сырья и биомассы» проводилась как сателлитное мероприятие V Международной научной школы-конференции молодых ученых «Катализ: от науки к промышленности». Организационный комитет выражает благодарность сотрудникам Томского государственного университета за помощь и содействие в организации работы школы молодых ученых.
В рамках проекта РНФ планируется проведение Третьей школы молодых ученых «Новые каталитические процессы глубокой переработки углеводородного сырья и биомассы» осенью 2019 года.
Материал подготовили
С.А. Яшник, С.С. Логунова, М.С. Суворова
Фото: Е.Н. Дубинина
(Институт катализа СО РАН, Новосибирск)
Заслуженный химик России,
Вице-президент Общественной организации
«Союз научных и инженерных общественных объединений»,
зам. Председателя химического общества
им. Менделеева Тульской области,
главный научный сотрудник НИАП-КАТАЛИЗАТОР,
профессор Е.З. Голосман
По инициативе специалистов по химической технологии создан Комитет Российского Союза научных и инженерных общественных объединений по проблемам энергоресурсо-эффективных химических технологий (ЭХТ). Одним из направлений работы Комитета будет совершенствование преподавания дисциплин инженерной химии, организация профессионально-общественной аккредитации образовательных программ и сертификации специалистов.
В конце 2018 года на базе Российского госуниверситета им. А.Н. Косыгина состоялось первое заседание Комитета ЭХТ. В нём приняли участие академики РАН, профессора, ректоры и проректоры вузов, директора предприятий и НИИ из Москвы, Иванова, Тамбова, Воронежа, Иркутска, Пятигорска, Казани, Белгорода, Уфы, Бийска, Вьетнама, Белоруссии, Великобритании и др. Было заслушано несколько докладов.
Наиболее интересным и глубоким был доклад «Химнефтегазпром: мировые тренды и российская действительность», который представил Президент международного института проблем химизации современной экономики профессор В.В. Бабкин.
На основе анализа мирового опыта по химизации экономики им было показано, что в мировой химической промышленности сохраняются высокие темпы роста ($4 трлн. в 2012г., а в 2030г. ожидается до $7,8 трлн.). При этом среднегодовой темп роста химического производства составит более 4,4% при среднегодовом темпе роста мирового ВВП на уровне 3%.
В 2017 году были импортированы десятки тысяч тонн катализаторов.

Слева – докладчик В.В. Бабкин
Напомню, что 85-90% продукции в химической и нефтехимической отрасли выпускается только с применением катализаторов. Вместе с тем ряд разработанных отечественных катализаторов не уступает лучшим мировым образцам. Например, срок службы катализаторов для процессов метанирования, получения защитных атмосфер и др., разработанных и выпускаемых НИАП-КАТАЛИЗАТОР составляет 15-25 лет, что обеспечивает заводам экономию в размере сотен миллионов рублей. Обидно, что ряд катализаторных производств в стране закрыт. В том числе самая крупная в Европе катализаторная фабрика в г. Дорогобуже, выпускавшая некогда тысячи тонн катализаторов метанирования, конверсии метана, синтеза анилина и других. Многие из них были разработаны в НИАП-КАТАЛИЗАТОР (Новомосковск) и ГИАП (Москва).
До сих пор в страну в большом количестве ввозятся средства защиты растений. Только в 2017 году было ввезено более 125000 тонн препаратов. А ведь есть свои! Помимо ранее выпускаемых в России средств защиты растений, специалистами НИАП-КАТАЛИЗАТОР и СФК АГРО были созданы новые препараты, например, МедьАГРО и др.
В Смоленске и Новомосковске налажен масштабный выпуск недорогого экологически чистого бифункционального (фунгицид + удобрение) медьсодержащего препарата защиты растений, превосходящего по своей эффективности такие известные фунгициды как медный купорос, бордосская смесь, купросат и др. В Смоленске ведется строительство специализированного крупного производства по выпуску препарата МедьАГРО.
Область применения нового препарата защиты растений весьма многообразна. После создания препарата «МедьАгро» в течение четырех лет проводились его испытания на дачных участках, в фермерских хозяйствах и крупных сельхозпредприятиях в Тульской, Смоленской, Тверской, Курганской, Челябинской и других областях, Краснодарском крае и в Крыму. В качестве объектов для агроиспытаний препаратом «МедьАгро» были обработаны яровая пшеница, ячмень, рапс, картофель, томаты, салаты, капуста, рис, виноград, плодово-ягодные культуры, деревья и кустарники. Кроме того, проводились испытания препарата по борьбе с паршой и лишайниками на плодовых деревьях. Препарат показал высокую эффективность.
Медный купорос и другие препараты после их нанесения на поверхность растений легко смываются дождем и загрязняют почву соединениями меди. В отличие от этих препаратов, Медь-Агро не корродирует оборудование и не закисляет почву, не вызывает ожоги растений. Раствор препарата может храниться без потери свойств длительное время (не менее 3-х лет). Приготовление рабочего раствора в отличие, например, от бордосской смеси, простое и технологичное. Высокая эффективность обработки растений достигается при минимальном расходе препарата на единицу обрабатываемой площади. Применение нового препарата защиты растений весьма многообразно.
После испытаний в нескольких НИИ и сельхозпредприятиях выявлена возможность применения модификаций препарата МедьАгро в качестве антисептика, например, при обработке древесины, а также как средства для профилактики и лечения рогатого скота от грибковых заболеваний.
Масштабное внедрение этого препарата тормозится длительным оформлением документов в различных госорганах. На оформление документации требуется несколько миллионов рублей. Видимо, проще импортировать дорогие зарубежные препараты. Безусловно, отказываться огульно от любой импортной продукции, от конкуренции не следует, но масштабы импортных поставок должны быть разумными и целесообразными.
К сожалению, отсутствует глобальная программа химизации индустрии страны и, конечно, ничтожны затраты на науку. По данным Президента института химизации профессора В.В. Бабкина, ежегодные расходы на НИОКР в химическом комплексе России в сравнении с другими развитыми странами ниже в сотни раз. Расходы на НИОКР в США составляют $10,7 млрд., в Китае – $10,4 млрд., в Японии – $7,7 млрд., а в России – $0,03 млрд.
А ещё относительно недавно (1965-1980 гг.) по масштабам и темпам химизации народного хозяйства наша страна была на первом месте в мире – в качестве примера можно привести строительство нескольких десятков гигантских агрегатов окисления аммиака. Примеры бурного строительства в химической и нефтехимической промышленности этого периода можно множить.
Были также заслушаны доклад профессора М.Г. Беренгартена «Инженерно-химическое образование – инновационные и/или традиционно-консервативные подходы» и другие сообщения ведущих специалистов.
К сожалению, за последние 25 лет престиж ученых и инженеров снизился. По этой причине, а также из-за недостаточного финансирования науки в последние годы десятки и десятки тысяч научных сотрудников, особенно молодых, уехали за границу. Потери для нашей страны колоссальные!
А мы все оптимизируем отраслевые институты (за 25 лет с карты страны из 6000 отраслевых НИИ и КБ исчезли более 5000). С 2013 года «реформируют» Академию наук. «Потерянное время» – так недавно в газете «Поиск» высказалась большая группа ученых о проблеме закрытия ряда вузов и реформировании РАН. Особенно болезненно это для регионов. Да, можно усилить несколько десятков университетов в Москве, Санкт-Петербурге, Новосибирске и других научных центрах. Конечно же, кто будет спорить, нужны хорошие институты с современной базой приборов и высококвалифицированными преподавателями. Да, хорошо бы все они были на уровне Массачусетского, Калифорнийского, Индийского... технологических институтов или нашего Физтеха. Но это же нереально.
На учебу в эти вузы приедут не более 15-20% подростков – остальных родные не смогут финансово обеспечить. По данным президента РАН и председателя комитета по науке Госдумы, почти 80% выпускников столичных вузов, приехавших на учебу из различных городов, остаются в мегаполисах. Кто же будет работать на заводах, в КБ, НИИ на российских просторах?
В Новомосковске в 1958 году был организован филиал Московского химико-технологического института (ныне НИ РХТУ). Еще недавно численность очных, заочных российских и иностранных студентов составляла около 5000 человек. Огромные корпуса, стадион, общежития. Выпускниками этого вуза (технологами, механиками, энергетиками, экономистами) в течение многих лет были обеспечены наши гиганты – НАК «Азот», «Проктер энд Гембл», «Гипс Кнауф», ГРЭС, «НИАП-КАТАЛИЗАТОР» и другие предприятия Новомосковска, Тулы, Ефремова, Щекино. Выпускники НИ РХТУ им. Д.И. Менделеева работают в десятках городов России. Сейчас в институте менее 1000 студентов. Финансирование настолько скудное, что несколько зимних периодов студенты и преподаватели на занятиях сидели в верхней одежде.

Чьи мудрейшие решения привели к резкому сокращению числа студентов и преподавателей? Еще немного, и город химиков, энергетиков, весь регион в целом останутся без химико-технологического вуза.
Похоже, что страна вернулась в тридцатые годы прошлого столетия, когда надо было в спешке готовить на рабфаках технических специалистов. Известно, что даже инженеров приходится доучивать на предприятиях применительно к задачам производства, отрасли. Нужны хорошие, полновесные инженеры. А вместо бакалавров (недоделанных инженеров) надо увеличить подготовку квалифицированных техников и лаборантов в техникумах и колледжах. При необходимости в вузах, испытывающих недобор абитуриентов, можно, на мой взгляд, готовить техников и лаборантов. Тем самым будет обеспечена и загрузка преподавателей этих вузов, и использование, как правило, более богатой, чем в техникумах и колледжах учебной базы. Если нужны в XXI веке высококвалифицированные рабочие, давайте выпускать их дополнительно в технических вузах. Напомню, что в развитых странах доля высококвалифицированных рабочих доходит до 50%, а в России составляет лишь 5-10%.
Давно обсуждается вопрос привлечения в вузы профессоров, доцентов и ведущих специалистов, работающих в НИИ и на промышленных предприятиях. Именно симбиоз вузовских преподавателей с такими совместителями обеспечивает высокую эффективность подготовки студентов и аспирантов.
К сожалению, в ряде вузов картина довольно печальная. При выполнении указа Президента о повышении зарплат работникам вузов идет вымывание совместителей, работающих на 0,1-0,5 ставки, тем самым как бы повышается средняя зарплата вузовских преподавателей. Для кого этот камуфляж? Лишаем студентов и аспирантов контактов с ведущими практическими специалистами. На президентском Совете по науке и образованию говорилось о потере популярности инженерного образования, нехватке квалифицированных инженеров, ставшей сдерживающим фактором развития экономики, и о необходимости приблизить образование к производству. О том, что студентов должны учить педагоги, знающие современное производство, что настоящий вуз – это вуз, где есть наука. Необходимо устранить барьеры, вроде запретов на совместительство. Какие же еще нужны указания?
Ныне от вузов и НИИ РАН требуют все большие объемы собственных разработок, внедренных в промышленность. И это чаще всего при отсутствии в них подготовленных специалистов, знающих промышленность, и крупных экспериментальных установок, без испытаний в которых практически невозможно или крайне рискованно начинать эксплуатацию разработки, прошедшей только лабораторные испытания. Нужны отраслевые институты.
Наш писатель, философ Петр Чаадаев говорил: «Я не могу любить родину с закрытыми глазами». Это было сказано более 200 лет тому назад. Любить страну, любить науку – значит решать реальные проблемы, а не талдычить о величии при явном отставании от многих технических прорывов, совершаемых в мире. И тем более это обидно при наличии блестящих научных умов и инженеров. Слой этот во всем мире достаточно тонок, а отъезд талантливых специалистов за рубеж, уход на работу в торговлю, в банки и прочие сферы губителен для страны.
В конце заседания состоялось обсуждение плана работы Комитета на 2019 год. Был рассмотрен вопрос об участии Комитета в подготовке Международного научно-технического симпозиума.
Сейчас необходимо в очередной раз вспомнить слова инициатора химизации нашей страны, министра химической промышленности, легендарного Леонида Костандова: «Какова химия – такова жизнь!»
The periodic table is an icon. But chemists still can’t agree on how to arrange it
New data about the elements and their relationships have led to debate over the 150-year-old table’s optimum ordering
One hundred fifty years after Russian chemist Dmitri Mendeleev published his system for neatly arranging the elements, the periodic table it gave birth to hangs in every chemistry classroom in the world and is one of the field’s most recognizable symbols. But the solid squares and familiar patterns of today’s table mask one of its fundamental characteristics: “the” periodic table does not exist.
It’s been mutable from the beginning. Not only has it grown as new elements have been discovered; it has also added columns and changed shape as we’ve gained new understanding of the elements’ properties and their relationships to one another. And scientists are still debating its optimum configuration.
Some believe chemical properties should dictate how the elements line up on the periodic table. Others think a more fundamental principle is needed, like electronic configuration or simply atomic number. Partisans are clashing over which elements belong in group 3, where helium should go, and how many columns the periodic table should have. They follow a long line of chemists and physicists who have worked and reworked the elements into a semblance of order.
“What I find interesting about the current debate is there are people who insist on there being one right table,” says Michael D. Gordin, a Princeton University historian who has written about Mendeleev, Julius Lothar Meyer, and other creators of early periodic tables. “It would have struck people like Mendeleev and Lothar Meyer as weird.” Gordin says the periodic table pioneers understood their tables to be a reflection of natural laws but recognized that different tables could represent those laws in different ways. That might be hard to imagine for those of us used to seeing the familiar shape of the table on our coffee mug or shower curtain.
Setting the table
Mendeleev wasn’t the first to recognize patterns in the elements, nor was he the first to try to depict those patterns in a diagram. Chemist Johann Wolfgang Döbereiner, for instance, identified triads of elements with shared properties in 1829. Today we’d recognize these as members of the same group or column of the periodic table, like chlorine, bromine, and iodine.
Geologist Alexandre-Émile Béguyer de Chancourtois published a kind of periodic table in 1862 in which the elements spiraled up a cylinder according to atomic weight. Each column of elements shared properties.
Mendeleev published his table, which he called a “periodic system,” in 1869. It included all 56 elements then known, and if you squint, it has a somewhat similar shape to the periodic table we see today, only tipped 90° on its side. Mendeleev arranged the elements in order of increasing weight and broke them into rows such that elements in each column shared valence, the number of other atoms they combined with, as well as other properties.
What made Mendeleev’s table special was his recognition that the periodic system was strong enough to predict undiscovered elements, which he left holes for, and even their properties. Lothar Meyer was independently working on an almost identical table, but Mendeleev beat him to publication by a few months and secured his place in history.
One can trace today’s controversies over how the periodic table should look to the discovery of quantum mechanics and atomic numbers. Mendeleev ostensibly organized his table by increasing atomic weight, but he gave chemical properties a deciding vote. For example, tellurium is slightly heavier than iodine, but Mendeleev put tellurium first because it has the same valence as oxygen, sulfur, and other elements in its group. Tables have retained that ordering. Mendeleev didn’t know that tellurium has one fewer proton—and thus is one atomic number less—than iodine, which explains why they each belong where they do. “When you get atomic number, it provides logic” to the periodic table, Gordin says.
Along with protons came the discovery of electrons and the quantum-mechanical idea of atomic orbitals. These findings provided a whole new kind of logic for the periodic system. Although the organization of Mendeleev’s system didn’t change, scientists could now see that it was electronic structure that largely dictated elements’ properties and explained why members of the same group were similar. The Madelung rule, or aufbau principle, that dictates that electrons fill the 1s orbital first and then the 2s and the 2p and so on, further explained how the elements were ordered.
That brings us to the tables we see today, which don’t look that different from the versions famed chemist Glenn T. Seaborg drew in the 1940s. Seaborg moved the f-block elements—also called the lanthanide and actinide series—out of the main table to leave them floating below. This decision is generally understood as a concession to convenience; if those elements were in line with the others, the table would be too wide to fit on a standard sheet of paper or the type would be too small to read.
Seaborg included 15 elements in his f-block. That doesn’t make a lot of sense from an electronic configuration point of view, since the f orbitals hold only 14 electrons. But many tables—including the table on the website of the International Union of Pure and Applied Chemistry (IUPAC), which has the last word on naming elements and molecules—share this feature. It’s a way of avoiding one of the most controversial questions about the periodic table: What elements belong in group 3? No one disputes scandium and yttrium. But which elements come below those two? Lanthanum and actinium? Or lutetium and lawrencium?
Group decision
Today there’s no standardization in the periodic tables found in classrooms, labs, and textbooks. Some avoid the group 3 question and use a 15-element f-block. Others put La and Ac in group 3, and still others have Lu and Lr, with the remainder of the f-block floating below.
IUPAC has convened a working group to make a definitive recommendation one way or the other. One motivation for forming the task group, according to IUPAC’s website, is to clear up confusion among students and teachers about which table is correct.
Philip Ball, a science writer and member of the working group, says the debate comes down to a fundamental question of whether physics or chemistry shapes the table. Put another way, he says the group is debating whether to side with the quantum physics that determines elements’ electronic configurations or with the way elements behave chemically.
On the side of chemical behavior is Guillermo Restrepo, a mathematical chemist at the Max Planck Institute for Mathematics in the Sciences. Restrepo takes a historical view of how the table should be organized. He points to Mendeleev and his contemporaries, who found their periodic systems by studying elements’ properties, often through their binding behavior. “At the core of the periodic system, what you have is chemistry,” Restrepo says, “and you need chemical reactions.”

Dmitri Mendeleev's first published table included all the known elements and left
space for the elements his periodic system predicted were still undiscovered.
Restrepo and colleagues analyzed some 4,700 binary compounds containing 94 elements to determine how chemical reactions inform the periodic system (MATCH Commun. Math. Comput. Chem.2012, 68,417). The molecules could consist of more than one atom but only two elements. The researchers created a map that groups elements near those that form similar compounds. For example, fluorine, chlorine, and the other halogens sit next to each other because they all bind to similar elements.
Restrepo says this similarity landscape shows that lanthanum is more similar to scandium and yttrium than lutetium is, so it should be in group 3. But the analysis doesn’t provide a good answer about Lr versus Ac. Restrepo says the problem is there isn’t much data on how Lr and Ac bind to other entities. While there are tens of thousands of compounds one can use to study the similarities of Sc, Y, La, and Lu, Ac provides only about 70 data points, and Lr, fewer than 40, according to Restrepo.
Eric Scerri, a philosopher of science at the University of California, Los Angeles, and the chair of the IUPAC task group, disagrees. He believes Sc, Y, Lu, and Lr should be the group 3 elements. Scerri thinks a focus on chemical or physical properties is misguided. He compares it to early botanists’ classification of flowers by their color or petal number.
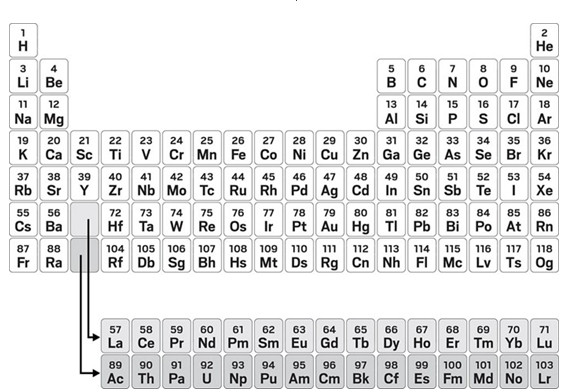
The periodic table on the website of the International Union of Pure and Applied Chemistry
avoids making a decision
about whether La and Ac or Lu and Lr are group 3 elements.
“You’ve got to go for something fundamental,” Scerri says, like electronic configuration. “Just to amass properties is never going to give you a definitive answer.”
Not that electronic configuration is perfect either, as Scerri will tell you. Exceptions have been made for some elements in the periodic table in terms of how their orbitals are filled, like copper. By the periodic table’s logic, all d-block elements should have filled s orbitals. But copper defies that logic. It should have the electron configuration [Ar] 3d9 4s2. Instead, its 4s atomic orbital remains unfilled, and one electron goes in its 3d shell, leading to the configuration [Ar] 3d10 4s1, which is more stable.
Scerri does prefer electronic configuration to what he calls “gross physical characteristics” for organizing the table. But he sees an even simpler logic to solve the group 3 problem: arrange by atomic number. If Scerri has his way, we’ll all have to get used to a newly arranged table.
Going wide
“My suggestion is simply this,” Scerri says. “Represent the periodic table in a 32-column format.”
Scerri calls 32 columns a more natural form for the periodic table and attributes the current dominance of 18 columns to convenience only. A 32-column table uses the atomic numbers as its logical foundation. Not only would it solve the group 3 question, but Scerri says 32 columns would be more correct because it puts the f-block in its rightful place: inside the table rather than floating below for convenience.
In a 32-column table arranged by ascending atomic number, lanthanum (atomic number 57) follows barium (number 56) to start the f-block, with actinium below it. That makes lutetium the first element in the third row of the d-block, with scandium and yttrium above it and lawrencium below to form group 3. Strict adherence to atomic number satisfies Scerri’s desire for a fundamental organizing principle and neatly sidesteps questions about chemical or physical properties.
Another table similar to the 32-column version has been proposed, but it uses electronic configuration rather than atomic numbers as its primary guide. French scientist Charles Janet’s left-step periodic table, devised in 1928, isn’t likely to make it into textbooks anytime soon, however. Janet moves the s-block to the right side of the table and includes helium at the top of group 2 because its s orbital, like other elements in that group, is filled.
Most tables place helium atop the noble gases. Scerri thinks helium’s demotion in the left-step table is one reason the table never got more traction, though recent experiments showing helium can form stable bonds help Janet’s argument that it belongs in a group with other reactive, rather than inert, elements.
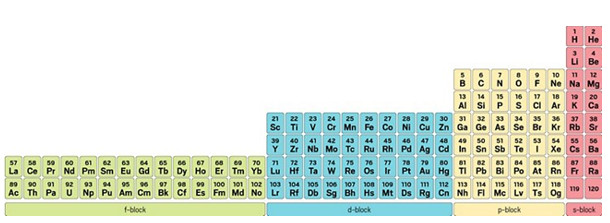
The left-step periodic table, invented by Charles
Janet, is strictly based on atomic orbitals and electron-filling
order.
Regardless, read from top to bottom and left to right, the left-step table more correctly conforms to the Madelung rule, which states that electrons must first fill the lowest-available electron levels before filling higher ones, Scerri says. It has a regularity that current tables don’t: two periods of 2 elements, two of 8 elements, two of 18, two of 32. And when element 121 is discovered, it will begin the g-block and two new periods of 50 elements.
Relative difficulty
The filling order that most chemists are used to might not hold up for much longer, however. Some calculations show the Madelung rule breaks down at higher atomic numbers because of relativistic effects. Electrons in large atoms move so fast that their behavior—and the properties of the atoms they belong to—begin to change.
Fans of relativistic effects will be happy to know there’s a table for them too. Pekka Pyykkö, a theoretical chemist at the University of Helsinki, calculated electron configurations up to element 172 and made a table for them (Phys. Chem. Chem. Phys. 2010, DOI: 10.1039/c0cp01575j). Pyykkö doesn’t bother with the group 3 question. His table leaves a hole under yttrium and has three 15-element rows in an f-block floating beneath the main table. Nor is he so constrained by atomic numbers. For instance, element 164 is followed by elements 139, 140, and then 169. According to Pyykkö’s calculations, 139 and 140 are the first elements with electrons in the 8p orbital.
This table is, of course, largely hypothetical. Scientists have not yet synthesized any elements beyond 118, and while several groups are working to do so, it’s possible we will reach the limits of our abilities to forge new elements well before element 172 or even 139.
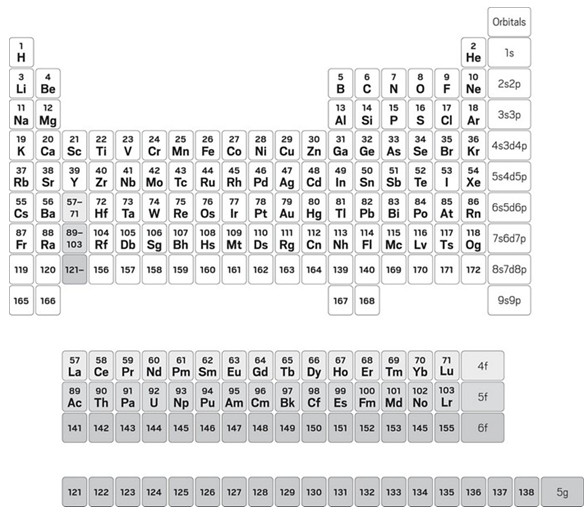
Pekka Pyykkö proposes a periodic table that goes all the way up to atomic number 172 and is based
on electronic configurations, which he calculated by taking relativistic effects into account.
“This is open land,” says Peter Schwerdtfeger, a theoretical chemist at Massey University studying superheavy elements. He calls Pyykkö’s calculations a “very good approximation” of the electron configurations but says more calculations are needed to pin down the precise characteristics of these elements. Pyykkö agrees. He too is waiting for more detailed calculations to show how wrong or right his table is.
And there are still other configurations of the elements that scientists are arguing for. Some tables look like rings or spirals. Some are 3-D, with lobes or stacks of element blocks. It seems less likely any of these will challenge the basic shape of the current table.
But Restrepo encourages scientists to think beyond just one table. “There’s a lot of discussion about if this table is good or bad, if this shape is better or worse,” he says. He prefers to focus on the periodic system rather than on the table. In a sense, he says, the system is like a sculpture, and the tables are shadows cast by lights shone from different angles. He says that allows chemists to find the periodic table or tables that are most useful to them, whether they’re looking for new elements or trying to understand properties in detail.
Gordin agrees on historical grounds. The periodic tables of the 19th century, he says, were made “to be flexible.” But he understands the resistance to radically different periodic tables, comparing new periodic tables to Pluto’s demotion from the ranks of planets. “The table you grew up with is the table you like.”
That raises a question about how much scientists and others should worry about these debates. “There’s a little group that argues over which [periodic table] is best,” Ball says. He believes the IUPAC group’s work is important because the current situation, with several competing tables, is confusing. But Ball says there’s no reason to think one table can capture the whole picture. “This notion of periodicity was so important for chemistry when the periodic table was first put forward and in the subsequent decades to make sense of this chaos of elements,” he says.
But today it should be more of a rule of thumb rather than a law of nature, Ball adds, arguing that there’s room for more than one periodic table: “Chemistry is about compromise.”
by Sam Lemonick
Chemical & Engineering News
January 7, 2019 (Vol. 97, issue 1)
|
January 29, 2019 IYPT2019 Opening Ceremony: the International Year of the Periodic Table, the 150th Anniversary of the Periodic Table of Chemical Elements Paris, France |
http://www.iypt2019.org |
|
18-20 февраля 2019 г. I Коршаковская Всероссийская конференция с международным участием «Поликонденсационные процессы и полимеры» Москва, Россия |
https://ineos.ac.ru/conferences/korshak-conf-2019 |
|
February
18-20, 2019 Hot-electron science and microscopic processes in plasmonics and catalysis – Faraday Discussion London, UK |
http://www.rsc.org/events/detail/27637/ hot-electron-science-and-microscopic-processes-in- plasmonics-and-catalysis-faraday-discussion |
|
February
25-27, 2019 3rd International Conference on Catalysis and Chemical Engineering (Catalysis-2019) Houston TX, United States |
https://unitedscientificgroup.com/conferences/catalysis |
|
March
11-13, 2019 3rd International Congress on Catalysis and Chemical Science Singapore, Singapore |
https://catalysiscongress.com |
|
18-21
марта 2019 г. XXXV Всероссийский симпозиум молодых ученых по химической кинетике Московская обл., Россия |
http://www.chem.msu.ru/rus/ChemKin |
|
April
8-10, 2019 Applied Catalysis & Chemical Engineering Dubai, United Arab Emirates |
https://www.eigenscientificgroup.com/conference/catalysis-2019 |
|
April
9-12, 2019 26th Croatian Meeting of Chemists and Chemical Engineers (26HSKIKI) Šibenik, Croatia |
http://www.26hskiki.org/en |
|
May
5-8, 2019 4th Green & Sustainable Chemistry Conference Dresden, Germany |
https://www.elsevier.com/events/conferences/green-and-sustainable-chemistry-conference |
|
May
13-15, 2019 Conference “Frontiers in Catalysis and Chemical Engineering” (EURO CAT 2019) London, UK |
https://www.longdom.com/eurocat |
|
May
13-17, 2019 International Symposium on Green Chemistry (ISGC) La Rochelle, France |
https://www.isgc-symposium.com |
|
May
15-17, 2019 5th International Conference on Polygeneration (ICP 2019) Fukuoka, Japan |
http://therme.mech.kyushu-u.ac.jp/ICP2019/ICP2019.html |
|
May
27-31, 2019 E-MRS Spring meeting – Symposium H: “Materials for applications in photocatalysis and photoconversion” Nice, France |
https://www.european-mrs.com/materials-applications-photocatalysis-and-photoconversion-emrs |
|
27-31
мая 2019 г. XI Всероссийская научная конференция с международным участием и школа молодых ученых «Химия и технология растительных веществ» Сыктывкар, Россия |
http://chemi.komisc.ru/ru/conference |
|
June
2-6, 2019 12th Natural Gas Conversion Symposium San Antonio, Texas, USA |
https://www.aiche.org/conferences/natural-gas-conversion-symposium/2019 |
|
June
2-6, 2019 14th International Symposium on Macrocyclic and Supramolecular Chemistry (ISMSC2019) Lecce, Italy |
https://ismsc2019.eu |
|
June
11-13, 2019 23rd Annual Green Chemistry & Engineering Conference and 9th International Conference on Green and Sustainable Chemistry Reston, VA (USA) |
http://gcande.org |
|
June
16-20, 2019 6th Nano Today Conference Lisbon, Portugal |
https://www.elsevier.com/events/conferences/nano-today-conference |
|
June
17-20, 2019 3rd International Conference on Applied Surface Science Pisa, Italy |
https://www.elsevier.com/events/conferences/international-conference-on-applied-surface-science |
|
June
19-23, 2019 22nd International Conference on Chemical Thermodynamics in Russia (RCCT 2019) St. Petersburg, Russia |
http://rcct2019.spbu.ru |
|
June
24-28, 2019 5th EuChemS Inorganic Chemistry Conference Moscow, Russia |
http://www.igic.ras.ru/docs/news/tcirkulyar_evrohim_2019.pdf |
|
June
26-30, 2019 6th European Conference on Environmental Applications of Advanced Oxidation Processes (EAAOP-6) Portoroz-Portorose, Slovenia |
http://eaaop6.ki.si |
|
July
5-12, 2019 50th General Assembly & 47th IUPAC World Chemistry Congress Paris, France |
https://www.iupac2019.org |
|
July
8-11, 2019 14th International Conference on Materials Chemistry (MC14) Birmingham, UK |
http://www.rsc.org/events/detail/31760/14th-international-conference-on-materials-chemistry-mc14 |
|
July
15-19, 2019 Catalysis Fundamentals and Practice Summer School University of Liverpool, UK |
https://www.liverpool.ac.uk/chemistry/events/catalysis-summer-school-2019 |
|
July
17-19, 2019 Energy Security and Chemical Engineering Congress 2019 (SChE 2019) Penang, Malaysia |
http://esche.ump.edu.my/index.php/en |
|
July
22-27, 2019 Summer School “Making Business with Green Chemistry & Sustainable Energy” Sarteano, Siena province, Italy |
www.eric-aisbl.eu/sarteano |
|
July
26-28, 2019 Mendeleev 150: 4th International Conference on the Periodic Table endorsed by IUPAC Saint Petersburg, Russia |
http://mendeleev150.ifmo.ru |
|
August
4-8, 2019 36th International Conference of Solution Chemistry Xining, China |
http://icsc2019.csp.escience.cn/dct/page/1 |
|
August
18-23, 2019 European Congress on Catalysis (14th EuropaCat) Aachen, Germany |
www.europacat2019.eu |
|
September
2-6, 2019 5th International Conference “Catalysis for Renewable Sources: Fuel, Energy, Chemicals” (CRS-5) Crete, Greece |
http://conf.nsc.ru/CRS5 |
|
September
2-6, 2019 1st International Conference on Noncovalent Interactions (ICNI) Lisbon, Portugal |
http://icni2019.eventos.chemistry.pt |
|
September
11-14, 2019 7th International Conference on Semiconductor Photochemistry Milano, Italy |
https://www.sp7.unimi.it |
|
September
23-27, 2019 5th International Congress on Catalysis for Biorefineries (Catbior 2019) Turku / Åbo, Finland |
http://www.catbior2019.fi |
|
October
7-11, 2019 XI International Conference “Mechanisms of Catalytic Reactions” (MCR-XI) Sochi, Russia |
http://conf.nsc.ru/mcr2019/en/ |
|
October
20-23, 2019 5th International Symposium on Innovative Materials and Processes in Energy Systems (IMPRES2019) Kanazawa, Japan |
http://www.knt.co.jp/ec/2019/impres2019/index.html |
|
|
|
|
June
28 – July 3, 2020 The World Conference on Carbon (CARBON 2020) Kyoto, Japan |
http://www.tanso.org/carbon2020 |
|
July
5-9, 2020 48th World Polymer Congress (MACRO2020) Jeju Island, Korea |
http://www.macro2020.org |
|
August
16-21, 2020 12th Triennial Congress of the World Association of Theoretical and Computational Chemists (WATOC 2020) Vancouver, Canada |
http://watoc2020.ca |


